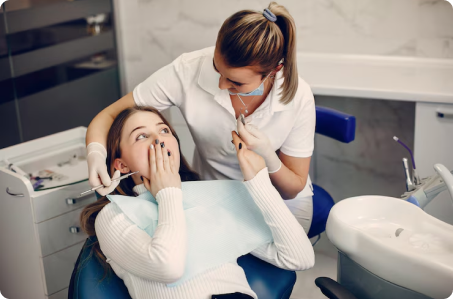
Tooth sensitivity affects many patients, causing discomfort and reduced quality of life. It can result from factors like micro-leakage, aging, and periodontal disease. The issues and benefits are described below.


Tooth sensitivity is a common problem that affects many people, particularly as they age. One of the primary causes of sensitivity is micro-leakage. The benefits of the technology are described below:

Blocks heat and cold sensitivity

Superior 3X bonding strength

Prolongs the life of dental restorations

Forms apatite to lock out micro-leakage

Contains botanical antimicrobial properties

Reduce Bacteria Growth

Enhances enamel remineralization

Long-lasting relief

Blocks heat and cold sensitivity

Superior 3X bonding strength

Prolongs the life of dental restorations

Forms apatite to lock out micro-leakage

Contains botanical antimicrobial properties

Reduce Bacteria Growth

Enhances enamel remineralization

Long-lasting relief

Advancing Oral Health Through Our Partnership

United in Delivering Cutting-Edge Dental Technology
Elevating Dental Practices Together with DentoClude™ F
At DentoClude™ F, we innovate dental solutions to enhance patient care. Our flagship product, DentoClude™ F, offers cutting-edge technology for effective tooth sensitivity relief.
© 2024, DentoClude™ F, All Rights Reserved, Powered by Kerlotech.

Are you a dental practitioner?